Conflict of interest
Authors are responsible for disclosing all financial and personal relationships between themselves and others that might be perceived by others as biasing their work. To prevent ambiguity, authors must state explicitly whether potential conflicts do or do not exist. COM, MG, AA, MEdlHA, SM, SS, ZK, MT, AS, AAH, MC: No conflict of interest; MS: Member of the Medigene Advisory Board, member of the MSD Advisory Board for Central and Eastern Europe and was member of the Auriga (ISDIN) Advisory Board, outside the submitted work; EN: Has received honorariums and grants from MEDA outside the submitted work.
Funding source
Medical writing support was funded by Meda Pharma S.p.A. a Mylan Company.
Abstract
Background
Anogenital warts (AGW) can cause economic burden on healthcare systems and are associated with emotional, psychological and physical issues.
Objective
To provide guidance to physicians on the diagnosis and management of AGW.
Methods
Fourteen global experts on AGW developed guidance on the diagnosis and management of AGW in an effort to unify international recommendations. Guidance was developed based on published international and national AGW guidelines and an evaluation of relevant literature published up to August 2016. Authors provided expert opinion based on their clinical experiences.
Results
A checklist for a patient's initial consultation is provided to help physicians when diagnosing AGW to get the relevant information from the patient in order to manage and treat the AGW effectively. A number of frequently asked questions are also provided to aid physicians when communicating with patients about AGW. Treatment of AGW should be individualized and selected based on the number, size, morphology, location, and keratinization of warts, and whether they are new or recurrent. Different techniques can be used to treat AGW including ablation, immunotherapy and other topical therapies. Combinations of these techniques are thought to be more effective at reducing AGW recurrence than monotherapy. A simplified algorithm was created suggesting patients with 1–5 warts should be treated with ablation followed by immunotherapy. Patients with >5 warts should use immunotherapy for 2 months followed by ablation and a second 2‐month course of immunotherapy. Guidance for daily practice situations and the subsequent action that can be taken, as well as an algorithm for treatment of large warts, were also created.
Conclusion
The guidance provided will help physicians with the diagnosis and management of AGW in order to improve the health and quality of life of patients with AGW.
Introduction
Anogenital warts (AGW) are epidermal growth lesions, caused by the different genotypes of human papillomavirus (HPV), which occur in the anogenital areas of males and females.1, 2More than 90% of cases of AGW are caused by HPV types 6 and 11.3, 4 Usually, HPV is contracted via sexual interactions, while other potential routes of viral transmission are rare.5 AGW represent a failure of immune recognition, although they only rarely have oncogenic potential and are not linked to cervical cancer.6 Although the transmission of HPV does not necessitate clinical lesions to be present, the viral burden of AGW is usually high and can therefore facilitate transmission.
Anogenital warts are a cosmetic nuisance and may cause substantial psychosocial issues for patients,6 as well as creating an economic burden on healthcare systems. The emotional and psychological issues associated with a diagnosis of AGW can include shame, embarrassment, anger, depression and guilt.4, 7, 8 Warts and the majority of the treatment modalities for the condition may also cause physical problems such as pain, itching, burning, irritation, and very rarely, obstruction during childbirth.2, 3 In addition, AGW can impact the sexual activity of patients, either through fear of transmission or embarrassment of lesions.9Furthermore, AGW are associated with substantial direct and indirect costs.10 A recent study estimated that the direct cost of genital wart management in the United Kingdom (UK) in 2012 was £58.44 million.11 The main drivers of cost were disease recurrence, the requirement for repeat physician visits and treatment.
The aim of this position statement is to provide guidance for physicians on the diagnosis and management of AGW in daily clinical practice. The guidance is intended to supplement, rather than replace, existing evidence‐based treatment guidelines.12-14
Methods
An international panel of 14 global experts on AGW was convened to develop guidance on the diagnosis and management of AGW in an effort to unify international recommendations. Guidance was developed based on a review of published international and national guidelines on AGW.12-14 A PubMed search was performed for articles published up to August 2016. Relevant literature on the diagnosis and management of AGW was evaluated. In situations where insufficient published information was available, recommendations were developed based on consensus of the authors' clinical experience. Professor O'Mahony led communications via email to discuss the development of the position statement and created the initial draft of the manuscript. The remaining 13 experts reviewed the manuscript and provided their input and clinical expertise. The experts provided all images included in the position statement.
Guidance for the diagnosis of AGW
In terms of diagnosis, the key challenge is ensuring that AGW are correctly identified. In the first instance, a diagnosis of AGW is usually made by the patient, which must then be confirmed by clinical inspection. In the case of uncertain lesions, polymerase chain reaction (PCR) diagnosis of different HPV genotypes can be attempted.
Typical presentations of AGW are shown in Fig. 1. AGW appear as papillomatous plaques or flat lesions and can be single or multiple in number. Lesions vary from flesh‐coloured to white, pink or brown.3 They typically manifest in areas of the body that are in close contact during sex: mainly on the anogenital areas such as vulva, penis, groin, perineum, perianal skin, but also in the oral cavity.2 Diagnosis of clinically typical AGW does not require histological confirmation.
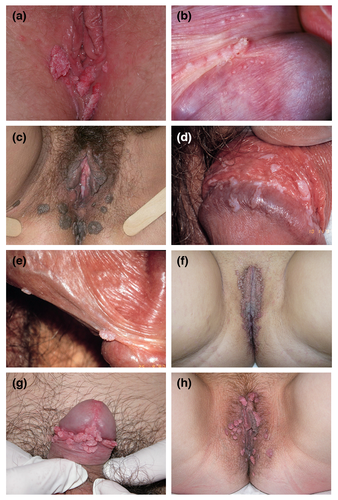
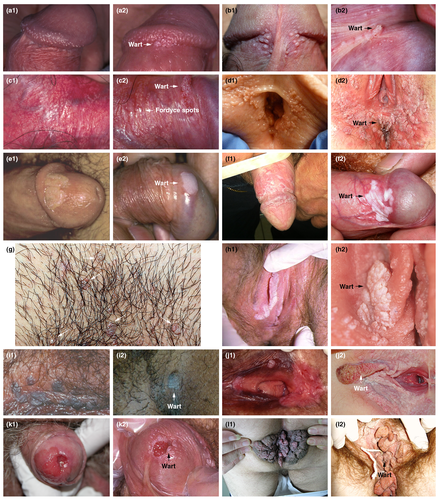
There are many conditions that can be misinterpreted as AGW (Fig. 2). Differential diagnoses that need to be excluded include normal skin variations (e.g. pearly penile papules, parafrenular glands, Fordyce spots, vestibular papillae, sebaceous cysts), other infectious or inflammatory conditions and other papules (syphilis on mucosal plates, molluscum contagiosum, lichen planus, psoriasis, condyloma lata) and benign or malignant neoplastic lesions (papillomatoses of vulva, nevi, verrucous carcinoma, invasive carcinoma, seborrhoeic keratosis, Bowen's disease, Buschke‐Löwenstein disease, pigmented or unpigmented grade 2–3 intraepithelial neoplasia, lymphangioma).2, 3 Pigmented or unusual lesions should be immediately referred to a specialist.
A checklist for the initial consultation with the patient is provided in Table 1. This will help physicians when diagnosing AGW to get the relevant information from the patient in order to manage and treat the AGW effectively. A number of questions that physicians are frequently asked are shown in Table 2, along with suggested answers. Patients should be reassured that if they have developed AGW, appropriate treatment can clear the warts within 3 months.5 Patients should be informed that AGW are of mostly sexual origin and are caused by HPV which is contagious; therefore, it is important for patients to disclose their AGW to recent sexual partners, who should be advised to visit a physician if they have developed AGW. Physicians should also inform patients that smokers have a 27% increased risk of developing AGW compared with non‐smokers.15 Furthermore, they should explain that HPV prevalence in patients who smoke is 48.2% compared with 37.5% for non‐smokers (P < 0.001).16 Generally, warts develop within weeks or months after acquiring HPV but in a significant number of cases, the virus can be dormant for months or years before warts emerge.17
| Checklist |
|---|
|
- AGW, anogenital warts.
| Questions | Answers |
|---|---|
| How did I get AGW? | AGW are caused by HPV.1 Usually, HPV is contracted via sexual interactions: indirect acquisition is rare5 |
| What is the risk of HPV transmission? | The risk of HPV transmission is very high (1.6 sexual interactions are enough to get the infection). The infection is very common and the vast majority of people have the virus during their lifetime |
| Is there a treatment? | Discuss the modalities and the limitations of treatment, explaining this will not eradicate the virus |
| Does smoking increase my risk of developing AGW? | Explain that smokers are at an increased risk of developing AGW and therefore, smoking cessation should be encouraged15 |
| How long will I have AGW for? | AGW can recur several times but with appropriate treatment, most warts should clear within 3 months5 |
| Is this the end of my sex life? | Reassure the patient that this is not the case |
| Should I disclose to my current and previous partner? | It is important to disclose you have AGW to your current partner in order to allow him/her to be checked |
| Should I always use a condom? | Explain that data have shown that increased levels of condom use is associated with increased clearance of HPV.88 It is therefore advisable to use condoms routinely |
| What are the risks during pregnancy? | AGW can become large during pregnancy5 but will usually disappear within weeks of delivery. In rare cases, HPV can be transmitted during child birth resulting in recurrent respiratory papillomatosis in the infant73, 78 |
| Will I develop cancer? | AGW are not related to cancer. AGW are caused by certain types of HPV, other types of HPV can cause cancer5 |
| Can AGW spread to other parts of the body? | It is very uncommon for AGW to spread to other body locations |
- AGW, anogenital warts; HPV, human papillomavirus.
Recommendations when selecting treatment options
Treatment should be individualized for each patient. Although untreated warts can resolve spontaneously,3, 17 most patients want an immediate intervention to eradicate them. Treatments need to be selected on the basis of considerations such as the number, size, morphology, location and keratinization of warts, and whether they are new or recurrent.8, 18 Wart area should be taken into consideration as one study showed that AGW with smaller surface areas (2–19 mm2) require significantly fewer treatment episodes and take less time to clear than those with larger surface areas (100–1038 mm2).19 Patient‐related considerations also need to be taken into account such as their preference for home or clinic‐based treatment, and the convenience of the regimen in terms of dosing frequency and duration.8, 18 Patient‐applied options are often preferred as they offer privacy, convenience and autonomy.18
Treatment options for AGW are provided in Table 3,20-64 and individual modalities are discussed in more detail below. A recent meta‐analysis of 18 studies of patient‐applied therapies concluded that all are more effective than placebo, although treatments cannot be ranked in terms of efficacy due to a lack of head‐to‐head comparisons.65
| Treatment | Mode of action | Schedule | Clearance rate (%) | Recurrence rate (%) | Advantages | Disadvantages | Refs | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ablative techniques | ||||||||||||
| Cryotherapy | Liquid nitrogen freezes and destroys lesions | Applied directly to lesions; repeat for two or three cycles | 46–96 | 18–39 |
|
| 20, 45-49 | |||||
| CO2 and Nd:YAG laser | Laser vaporizes lesions | Under local anaesthesia, protocol depends on type of laser | 23–95 | 2.5–77 |
|
| 20, 48, 50 | |||||
| Electrocautery | High‐frequency electrical currents cause thermal damage to infected tissue | Under local anaesthesia, base of lesion excised; repeat as required | 35–94 | 20–25 |
|
| 18, 20, 49, 51 | |||||
| Surgery | Scissor or scalpel excision | Under local or general anaesthesia; base of lesion excised | 89–93 | 18–65 |
|
| 52-54 | |||||
| Trichloroacetic acid (33–50%) | Acid induces a chemical burn | One to three times per week; repeat as necessary | 70–100 | 18–36 |
|
| 20, 45, 47, 55 | |||||
| Immunotherapies | ||||||||||||
| Imiquimod 5% | Immunomodulator: stimulates interferon and cytokine production | Three nights per week for up to 16 weeks or longer | 35–75 | 6 |
|
| 20-26, 41, 56-62 | |||||
| Imiquimod 3.75% | Immunomodulator: stimulates interferon and cytokine production | Once daily before bedtime for up to 8 weeks | 19–37 | 15–19 |
|
| 20, 27-29 | |||||
| Sinecatechins 10% and 15% | Inflammatory response modulator | Three times daily for up to 16 weeks | 40–81% | 7–12 |
|
| 20, 30-34 | |||||
| Other topical therapy | ||||||||||||
| Podophyllotoxin 0.5% (alcoholic solution) 0.15% (cream) | Antimitotic agent induces tissue necrosis | Twice‐daily to affected areas for 3 consecutive days per week; discontinue for 4 days; repeat for up to 4 weeks | 45–94 | 11–100 |
|
| 20, 31, 35-40, 42-44, 63 | |||||
| Nitric–zinc complex topical solution | Induces a caustic effect on the wart through mummification and protein denaturation/coagulation action | Once or up to four times; repeat at 2‐week intervals if needed | 90–99 | Not evaluated |
|
| 64 | |||||
- AGW, anogenital warts.
Ablative techniques
Ablative techniques are commonly used by physicians to remove warts in daily practice. However, most are awkward and painful for the patient. The major frustration is the high rate of recurrence with these treatments (see below) and the need for repeat therapeutic interventions. Ablative techniques are associated with a risk of bleeding, tissue destruction, slow wound healing and scarring.44, 66
Cryotherapy
Cryotherapy is the freezing of AGW using liquid nitrogen and is often used at a patient's first clinic visit to help initiate removal of the AGW. Various handheld devices, such as Hydrozid®(Dunelm Pharmaceuticals, Drogheda, Ireland), as well as cryotherapy machines can be used for the procedure. Hydrozid® is a disposable canister, which can be sprayed accurately onto the wart (Fig. 3). This treatment option can be repeated weekly, biweekly or every 3 weeks and is a relatively simple, inexpensive technique, requiring minimal training. However, it requires many clinic visits and a second or third cycle of freezing may be needed. Clearance rates of 46–96% have been reported although treatment can cause pain, necrosis and blistering.45-49, 66 For non‐Caucasians, post‐inflammatory hypo/hyperpigmentation after treatment with cryotherapy can be frustrating; therefore, this should be discussed with patients before proceeding with this treatment option.
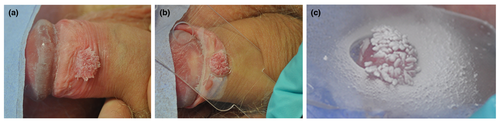
Carbon dioxide and Nd:YAG laser
Carbon dioxide (CO2) and Nd:YAG lasers vaporize lesions using focused infrared light energy; however, it is not always possible to know the extent of the infected tissue, and therefore, vaporizing large regions around the warts is not always feasible. Local anaesthesia is usually required, especially on extensive and thick lesions as it can penetrate deeply into the lesions.50
This treatment option is used less frequently than other therapies as it requires specialized and costly equipment, and has an increased risk of serious complications unless used by an experienced physician.1 However, clearance rates of up to 95% have been reported in clinical studies, with a head‐to‐head comparison showing greater efficacy than cryotherapy.47, 48, 50 It is important to note that fumes from laser treatment contain contagious particles and adequate measures should be taken to prevent the virus from spreading. Masks and smoke evacuators should therefore be used.
Electrocautery
Electrocautery uses high‐frequency electrical currents to destroy AGW and requires local anaesthesia and physician expertise.18, 20 Clinical studies have shown clearance rates of 35–94%.20, 49, 51 As fumes from electrocautery contain contagious particles, preventative measures should be put in place to stop the virus spreading.
Surgery
Surgery is performed using scissors or a scalpel and is particularly suited for removing large lesions causing obstruction. Local or general anaesthesia is required, and patients may experience post‐operative pain.50 Clearance rates of up to 93% have been reported in clinical studies.51-53
Trichloroacetic acid (TCA; 33–50%)
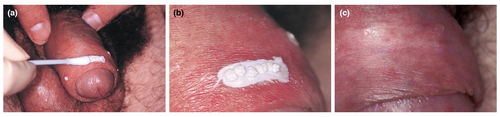
Physician‐applied acidic treatment causes a chemical burn that destroys the AGW. The acid can be administered up to three times per week until the warts have cleared. This process requires a skilled professional to choose the appropriate lesion and duration of application but it is easy to apply and effective for treating AGW, with clearance rates of 70–100% reported in clinical studies (Fig. 4). However, side‐effects such as local discomfort, burning and ulceration are common, hence the need for careful application.45-47, 53, 55, 66 TCA can also be used to treat small lesions; however, it is not frequently used due to a high recurrence rate and the risk of side‐effects.
Immunotherapies
Immunotherapies use stimulation of the body's own immune system to clear infected lesions.
Imiquimod 5% or 3.75%
Imiquimod is an immune response modifier with antiviral activity. This Toll‐like receptor 7 agonist induces the production of cytokines, which enhance the ability of antigen presenting cells to present viral antigens to reactive T lymphocytes.21, 22, 65 Imiquimod 5% has been approved for the treatment of AGW worldwide, whereas imiquimod 3.75% is only approved in certain countries such as the United States of America (USA) and Canada. Imiquimod 5% is self‐applied by the patient three nights per week for up to 16 weeks; if no improvement has occurred after 4–6 weeks, treatment can be applied daily. In comparison, imiquimod 3.75% is self‐applied once‐nightly for up to 8 weeks. Imiquimod 5% may be applied for longer durations if there is a good clinical result but complete clearance has not occurred at the end of the initial treatment period.28 Both imiquimod formulations are associated with local skin reactions such as erythema, pruritus, burning, pain and sometimes erosions. These are all signs that the immune system has been activated. The lower concentration of imiquimod in the 3.75% cream is associated with improved tolerability. In addition, the shorter treatment duration and dosing simplicity may improve patients' adherence to the regimen.27 AGW clearance rates from clinical studies range from 35 to 75% with the 5% cream20, 23, 24, 41, 56-62 and 19 to 37% with the 3.75% formulation,20, 27-29 with higher clearance rates in women than men.29 Further studies have shown that patients find imiquimod 5% to be both acceptable and preferable to other AGW treatments. A study of 559 patients with AGW reported excellent, very good or good with imiquimod 5% in 27.4%, 36.1% and 23.0% of patients, respectively.25 In addition, a survey of 629 patients showed that imiquimod 5% was rated better in terms of overall satisfaction, convenience, time to clearance and lack of associated pain than other AGW therapies.26
Sinecatechins
Sinecatechins consist of green tea polyphenols, which have anti‐inflammatory, anti‐proliferative, pro‐apoptotic and antiviral properties, although their exact mode of action is unknown.30, 31 They are available for the treatment of AGW as a 10% and 15% ointment or cream, which is self‐applied by the patient three times per day for a maximum of 16 weeks.31 In comparison, imiquimod 5% is applied three times weekly while application of imiquimod 3.75% is once daily.30 Patient adherence to dosing regimens should be considered, as compliance is important in achieving treatment effectiveness.30 An additional factor that may affect compliance is that sinecatechin 15% ointment is a brown formulation,67 which could stain light‐coloured clothing and bedding, reducing patient adherence.
Clinical studies of sinecatechins have shown similar clearance rates to that of imiquimod 5% therapy. Sinecatechins have resulted in complete clearance rates of 40–81%, with comparable differences in response rates between the 10% and 15% ointments.31-34Furthermore, the recurrence rate with sinecatechin 10% ointment was 6.8% after 12 weeks of treatment68 and 12% with sinecatechin 10% cream following 12 weeks of treatment.32This was higher than the recurrence rate of 6.2% observed with imiquimod 5% treatment after 3 months and 6.3% at 6 months.62 No significant difference in clearance or recurrence rates has been found between sinecatechin 10% cream and placebo.30 No long‐term data are available for sinecatechins. The most commonly observed application site reactions are erythema, pruritus, irritation, pain and ulceration; these side‐effects may indicate the greater likelihood of a clinical response.30
Other topical therapies
Podophyllotoxin 0.15% cream or 0.5% alcoholic solution
Podophyllotoxin stops division of infected cells causing tissue necrosis.20, 31, 35 It can be self‐applied by patients twice‐daily for three consecutive days, separated by a 4‐day treatment‐free period and repeated for up to 4 weeks. Patients need to carefully apply the solution to the lesions and avoid contact with healthy skin. Clearance rates from clinical studies range from 45 to 94%, with common side‐effects including pain, itching, burning, erosion and inflammation.20, 31, 35-40, 42-44, 63
Nitric zinc
Nitric–zinc complex is a solution for topical application containing nitric acid, zinc, copper and organic acids, currently used to treat common warts.64 It has a caustic effect on the wart through mummification and protein denaturation or a coagulation action.64 The solution can be applied topically once, or up to four times, at 2‐week intervals until a complete clinical cure rate is observed.64 Clearance rates in one study ranged from 90 to 99%, and the product was well tolerated with no serious adverse events recorded.64 Initial data suggest promising efficacy in AGW; however, additional studies are needed.
Guidance for preventing the recurrence of AGW
Anogenital warts recurrence is common and frustrating for patients and physicians.18Recurrence rates with conventional ablative techniques are relatively high (Table 3), since these methods only remove the visible wart without affecting the underlying HPV infection.44, 66 Of currently available treatments, recurrence rates are very low with immunotherapies, imiquimod (6–19%)20, 23, 28, 41, 56, 57, 60, 62 and sinecatechins (4–12%),32-34 as these treatments stimulate the host's immune response to clear the warts.
Studies have shown that a combination of ablative techniques followed by immunotherapy may lead to even lower recurrence rates; ablation provides rapid clearance but has high recurrence rates while immunotherapy has slow clearance rates and a lower risk of recurrence.20, 69 A study of 211 patients showed that imiquimod 5% applied within 3 weeks after laser therapy (to ensure complete wound healing) was associated with a low rate of wart recurrences of 11.8% over 6 months of follow‐up.69 Results of a 3‐arm, open‐label study involving 358 patients showed that 6‐month recurrence rates in those randomized to a combination of ablation followed by imiquimod 5% (8%) were lower than those after ablation alone (26%), but similar to imiquimod 5% monotherapy (6%).62 Furthermore, the results of a retrospective case series of 27 patients showed that combined treatment with cryotherapy, podophyllin 25% and subsequent use of sinecatechins 15% ointment led to a recurrence rate of 7.4% after 6 months of follow‐up.70 Gilson et al.,71 further showed that a combination of cryotherapy and podophyllotoxin cream 0.15% resulted in a higher clearance rate (60%) than with cryotherapy alone (45.7%) at both 4 and 12 weeks. However, these differences were not statistically significant.71
Pre‐treatment of AGW with imiquimod to stimulate an immune reaction followed by surgery is also associated with low recurrence rates. A retrospective study of 60 patients with anogenital warts showed that the recurrence rate during long‐term follow‐up (up to 7 years) was lower for patients with complete responses to imiquimod 5% monotherapy (15%), or with surgical removal of residual warts after imiquimod 5% (20%), compared with surgery alone (65%).54
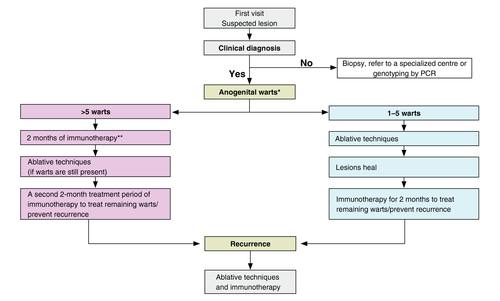
A simplified algorithm for AGW treatment
A new simplified treatment algorithm for AGW is shown in Fig. 5. Patients with a confirmed clinical diagnosis of AGW are initially classified by their number of warts. Patients with 1–5 warts may be treated in the first instance with ablation. Once the lesions have healed, immunotherapy can be used for 2 months to treat remaining warts and/or prevent recurrence. The choice of ablative technique is at the discretion of the physician taking factors such as the location of the wart into consideration. For those with more than five warts, the expert's recommendation is to pre‐treat the AGW with an immunotherapy for 2 months to see whether an immune response can be stimulated. If the warts are still present following this treatment, an ablative technique can be used to remove the AGW. It is recommended to use a second 2‐month course of immunotherapy to treat remaining warts and/or prevent recurrence. It is recognized that there are many different algorithms for the treatment of AGW and that the choice is dependent on many factors. For example, if all staff are experienced in ablative techniques, then irrespective of the number of warts, the clinic protocol may dictate that ablation is used on all patients with warts at first visit, followed by immunotherapy. In the UK, this is usual practice as it is preferable for a reduced number of clinic visits.
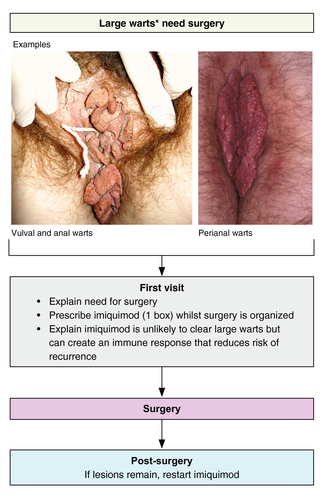
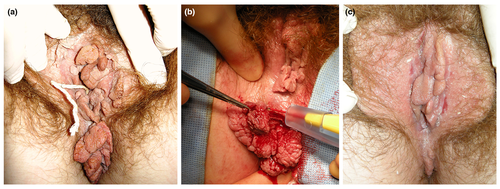
An algorithm for the treatment of patients with large AGW is also shown in Fig. 6. Large warts are defined as too large for local TCA or cryotherapy, and patients with these warts should be referred to a specialist. Based on clinical experience, our recommendation is to initially pre‐treat the AGW with immunotherapy for up to 16 weeks to stimulate an immune reaction to reduce the risk of recurrence. In support of this, long‐term recurrence rates are lower for patients pre‐treated with imiquimod 5% followed by surgery compared with surgery alone.54, 70 Evidence for other immunotherapies in this setting is not currently available. The AGW should then be surgically removed under general anaesthesia, with immunotherapy being re‐started if there are residual or recurrent lesions. A histological examination of the excised tissue should be performed to exclude verrucous or squamous cell carcinoma. An example of a patient treated with this approach is shown in Fig. 7.
Guidance for daily practice situations
Guidance for daily practice situations and the subsequent action that can be taken are shown in Table 4.3, 8, 12, 16, 23, 26, 30, 50, 54, 56, 62, 66, 72-80
| Daily practice situations | Actions |
|---|---|
| AGW remaining following ablation |
|
| Experience or fear of local side‐effects in genital area |
|
| Limited initial efficacy with imiquimod | |
| Lack of adherence |
|
| Lumps left may not be true warts |
|
| Heavy cigarette smoking | |
| Pregnancy |
|
| Immune suppression |
|
| Other conditions (i.e. diabetes, eczema, psoriasis) |
|
| Concomitant local infections (e.g. bacterial, fungal etc.) |
|
Preventing AGW
Anogenital warts can now be effectively prevented using the quadrivalent (HPV 6, 11, 16 and 18) or nanovalent (HPV 6, 11, 16, 18, 31, 33, 45, 52 and 58) HPV vaccines; these protect against HPV types that cause AGW, cervical cancer and other types of anogenital and oral cancer. The HPV quadrivalent vaccine has shown to be up to 100% effective in preventing AGW in association with vaccine‐type HPV in women.81, 82 After its introduction in Australia, a study with a 4‐year follow‐up showed a 59% reduction in the prevalence of AGW in young females.83 There was also a concomitant, although less marked, decline in AGW in heterosexual men following introduction of the vaccine.83, 84 Prevention of AGW with the HPV vaccine could therefore result in substantial savings in healthcare costs and reduction in workload for sexual health clinics.85 The vaccine is also effective in 12‐ to 15‐year‐old boys and is licensed for use in both sexes in most countries where it is available.85, 86 Evidence on whether the vaccination could be useful in AGW treatment is not yet clear; however, there are scientific data supporting use of the vaccination in individuals previously exposed to HPV.87
Conclusions
The guidance provided will help physicians with the diagnosis and management of AGW in daily clinical practice, in order to improve the health and quality of life of patients with AGW. The suggested therapeutic approach is flexible, allowing physicians to choose treatment depending on local availability and physician expertise, as well as considering patient preferences.
Acknowledgements
The authors were assisted in the preparation of the manuscript by David Harrison, Medscript Ltd, and Laura Brennan, a professional medical writer at CircleScience, an Ashfield Company, part of UDG Healthcare plc. Medical writing support was funded by Meda Pharma S.p.A. a Mylan Company. The authors would like to thank Professor Parent (Department of Dermatology, Hôpital Erasme, Université Libre de Bruxelles, Brussels, Belgium) for contributing photographs to this manuscript.
References
- 1 . Warts (genital). BMJ Clin Evid 2010; 2010: 1602.
- 2 , , et al. Clinical features of external genital warts. J Cutan Med Surg 2013; 17( Suppl 2): S55– S60.
- 3 . New approach to managing genital warts. Can Fam Physician 2013; 59: 731– 736.
- 4 , , et al. Understanding genital warts: epidemiology, pathogenesis, and burden of disease of human papillomavirus. J Cutan Med Surg 2013; 17( Suppl 2): S47– S54.
- 5 , . Frequently asked questions about genital warts in the genitourinary medicine clinic: an update and review of recent literature. Sex Transm Infect 2008; 84: 3– 7.
- 6 . Genital warts: current and future management options. Am J Clin Dermatol 2005;6: 239– 243.
- 7 , , et al. Psychological burden of anogenital warts. J Eur Acad Dermatol Venereol 2009; 23: 1035– 1038.
- 8 , , et al. Managing external genital warts: practical aspects of treatment and prevention. J Cutan Med Surg 2013; 17( Suppl 2): S68– S75.
- 9 , . Follow‐up of men with genital papilloma virus infection. Psychosexual aspects. Acta Derm Venereol 1992; 72: 185– 186.
- 10 , , et al. Economic and humanistic burden of external genital warts. Pharmacoeconomics 2012; 30: 1– 16.
- 11 , , et al. The costs of managing genital warts in the UK by devolved nation: England, Scotland, Wales and Northern Ireland. Int J STD AIDS 2016; 27: 51– 57.
- 12 , , et al. 2012 European guideline for the management of anogenital warts. J Eur Acad Dermatol Venereol 2013; 27: e263– e270.
- 13 , , et al. UK national guidelines on the management of anogenital warts 2015. URL http://www.bashh.org/documents/UK%20national%20guideline%20on%20Warts%202015%20FINALpdf. (last accessed: 26 February 2018).
- 14 Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines: Anogenital warts. 2015 URL http://www.cdc.gov/std/tg2015/warts.htm. (last accessed: 26 February 2018)
- 15 , , et al. Association between smoking and genital warts: longitudinal analysis. Sex Transm Infect 2010; 86: 258– 262.
- 16 , , . The impact of smoking on HPV infection and the development of anogenital warts. Int J Colorectal Dis 2014; 29: 899– 908.
- 17 , , . Genital warts: a comprehensive review. J Clin Aesthet Dermatol 2012; 5: 25– 36.
- 18 , , et al. Therapeutic options for external genital warts. J Cutan Med Surg 2013; 17( Suppl 2): S61– S67.
- 19 , , . Factors involved in clearance of genital warts. Int J STD AIDS2001; 12: 789– 792.
- 20 , , et al. Sinecatechins and imiquimod as proactive sequential therapy of external genital and perianal warts in adults. Int J STD AIDS 2017; 28: 1433– 1443.
- 21 . Immunomodulatory and pharmacologic properties of imiquimod. J Am Acad Dermatol 2000; 43: S6– S11.
- 22 . Mechanism of action and other potential roles of an immune response modifier. Cutis 2007; 79: 36– 45.
- 23 , , et al. Imiquimod, a patient‐applied immune‐response modifier for treatment of external genital warts. Antimicrob Agents Chemother 1998; 42: 789– 794.
- 24 , , et al. Efficacy and safety of imiquimod versus podophyllotoxin in the treatment of anogenital warts. Sex Transm Dis 2011; 38: 216– 218.
- 25 , . Effectiveness, satisfaction and compliance with imiquimod in the treatment of external anogenital warts. Int J STD AIDS 2003; 14: 11– 17.
- 26 , , et al. New patient‐applied therapy for anogenital warts is rated favourably by patients. Int J STD AIDS 2001; 12: 565– 570.
- 27 , . The role of imiquimod 3.75% cream in the treatment of external genital warts. Skin Therapy Lett 2012; 17: 5– 7.
- 28 , , et al. Imiquimod 3.75% cream applied daily to treat anogenital warts: combined results from women in two randomized, placebo‐controlled studies. Infect Dis Obstet Gynecol 2011; 2011: 806105.
- 29 , , . Imiquimod cream 2.5% and 3.75% applied once daily to treat external genital warts in men. Cutis 2015; 96: 277– 282.
- 30 , . Sinecatechins 10% ointment: a green tea extract for the treatment of external genital warts. Skin Therapy Lett 2015; 20: 6– 8.
- 31 , . Sinecatechins (Polyphenon E) ointment for treatment of external genital warts and possible future indications. Expert Opin Biol Ther 2014; 14: 1033– 1043.
- 32 , , et al. A randomized, double‐blind, four‐arm parallel‐group, placebo‐controlled Phase II/III study to investigate the clinical efficacy of two galenic formulations of Polyphenon E in the treatment of external genital warts. J Eur Acad Dermatol Venereol 2007; 21:1404– 1412.
- 33 , , et al. Topical Polyphenon E in the treatment of external genital and perianal warts: a randomized controlled trial. Br J Dermatol 2008; 158: 1329– 1338.
- 34 , , et al. Sinecatechins, a defined green tea extract, in the treatment of external anogenital warts: a randomized controlled trial. Obstet Gynecol 2008; 111:1371– 1379.
- 35 , . Management of external genital warts. Am Fam Physician 2014; 90:312– 318.
- 36 , , et al. A single‐blind study of podophyllotoxin cream 0.5% and podophyllotoxin solution 0.5% in male patients with genital warts. Genitourin Med 1995; 71:391– 392.
- 37 , , et al. Self‐treatment of female external genital warts with 0.5% podophyllotoxin cream (Condyline) vs weekly applications of 20% podophyllin solution. Int J STD AIDS 1995; 6: 257– 261.
- 38 , , et al. An open, comparative, study of the efficacy of 0.5% podophyllotoxin lotion and 25% podophyllotoxin solution in the treatment of condylomata acuminata in males and females. Int J STD AIDS 1993; 4: 194– 199.
- 39 , . Self‐treatment using a 0.5% podophyllotoxin cream of external genital condylomata acuminata in women. A placebo‐controlled, double‐blind study. Sex Transm Dis 1992;19: 170– 174.
- 40 , , et al. A double‐blind, randomized trial of 0.5% podofilox and placebo for the treatment of genital warts in women. Obstet Gynecol 1991; 77: 735– 739.
- 41 , , et al. Self‐administered topical 5% imiquimod cream for external anogenital warts. HPV Study Group. Human PapillomaVirus. Arch Dermatol 1998; 134: 25– 30.
- 42 , , et al. Self‐treatment using 0.25–0.50% podophyllotoxin‐ethanol solutions against penile condylomata acuminata: a placebo‐controlled comparative study. Genitourin Med 1994; 70: 105– 109.
- 43 , , et al. Patient‐applied podofilox for treatment of genital warts. Lancet 1989; 1: 831– 834.
- 44 , , et al. Double‐blind randomized clinical trial of self‐administered podofilox solution versus vehicle in the treatment of genital warts. Am J Med 1990; 88: 465– 469.
- 45 , , et al. Cryotherapy compared with trichloroacetic acid in treating genital warts. Genitourin Med 1987; 63: 390– 392.
- 46 , . Cryotherapy in the treatment of condylomata acuminata: a controlled study of 64 patients. J Dermatol Surg Oncol 1991; 17: 273– 276.
- 47 , , . Treatment of external genital warts comparing cryotherapy (liquid nitrogen) and trichloroacetic acid. Sex Transm Dis 1993; 20: 344– 345.
- 48 , , . CO(2) Laser therapy versus cryotherapy in treatment of genital warts; a Randomized Controlled Trial (RCT). Iran J Microbiol 2012; 4: 187– 190.
- 49 , , et al. Treatment of external genital warts: a randomised clinical trial comparing podophyllin, cryotherapy, and electrodesiccation. Genitourin Med 1990; 66:16– 19.
- 50 , . An evidence‐based review of medical and surgical treatments of genital warts. Dermatol Online J 2006; 12: 5.
- 51 , , et al. Randomized clinical trial comparing systemic interferon with diathermocoagulation in primary multiple and widespread anogenital condyloma. Obstet Gynecol 1989; 74: 393– 397.
- 52 . Comparison of podophyllin application with simple surgical excision in clearance and recurrence of perianal condylomata acuminata. Lancet 1985; 2: 1146– 1148.
- 53 . Podophyllin versus scissor excision in the treatment of perianal condylomata acuminata: a prospective study. Br J Surg 1989; 76: 1067– 1068.
- 54 , , . Treatment of anogenital warts with imiquimod 5% cream followed by surgical excision of residual lesions. J Am Acad Dermatol 2002; 47: S212– S216.
- 55 , , et al. Therapeutic value of trichloroacetic acid in the treatment of isolated genital warts on the external female genitalia. J Reprod Med 2007; 52: 521– 525.
- 56 , , et al. Treatment of genital warts with an immune‐response modifier (imiquimod). J Am Acad Dermatol 1998; 38: 230– 239.
- 57 , , et al. Imiquimod 5% cream is a safe and effective self‐applied treatment for anogenital warts–results of an open‐label, multicentre Phase IIIB trial. Int J STD AIDS 2001; 12: 722– 729.
- 58 , , et al. Safety and efficacy of imiquimod 5% cream in the treatment of penile genital warts in uncircumcised men when applied three times weekly or once per day. Int J STD AIDS 2001; 12: 22– 28.
- 59 , , et al. Treatment of external genital warts in men using 5% imiquimod cream applied three times a week, once daily, twice daily, or three times a day. Sex Transm Dis 2001; 28: 226– 231.
- 60 , , et al. Topical imiquimod 5% cream in external anogenital warts: a randomized, double‐blind, placebo‐controlled study. J Dermatol 2004; 31: 627– 631.
- 61 , , et al. An open‐label phase II pilot study investigating the optimal duration of imiquimod 5% cream for the treatment of external genital warts in women. Int J STD AIDS 2006; 17: 448– 452.
- 62 , , et al. Randomized, comparative trial on the sustained efficacy of topical imiquimod 5% cream versus conventional ablative methods in external anogenital warts. Eur J Dermatol 2006; 16: 642– 648.
- 63 , , . Podophyllotoxin 0.5% v podophyllin 20% to treat penile warts. Genitourin Med 1988; 64: 263– 265.
- 64 , , et al. Efficacy and tolerability of nitric‐zinc complex in the treatment of external genital warts and "difficult‐to‐treat" warts: a "proof of concept", prospective, multicentre, open study. G Ital Dermatol Venereol 2015; 150: 643– 648.
- 65 , , et al. Self‐administered interventions for anogenital warts in immunocompetent patients: a systematic review and meta‐analysis. Sex Transm Infect2017; 93: 155– 161.
- 66 . Evaluation of imiquimod for the therapy of external genital and anal warts in comparison with destructive therapies. Br J Dermatol 2007; 157( Suppl 2): 52– 55.
- 67 . Update on the treatment of genital warts. Dermatol Online J 2013; 19: 18559.
- 68 , , et al. Polyphenon E: a new treatment for external anogenital warts. Br J Dermatol 2010; 162: 176– 184.
- 69 , , et al. Effect of adjuvant imiquimod 5% cream on sustained clearance of anogenital warts following laser treatment. Infect Dis Obstet Gynecol 2002;10: 79– 88.
- 70 , , et al. Combined treatment of anogenital HPV infection with cryodestruction, podophyllin 25% and post‐ablation immunomodulation with sinecatechins 15% ointment – a retrospective analysis. Int J STD AIDS 2016; 27: 1071– 1078.
- 71 , , et al. A multicentre, randomised, double‐blind, placebo controlled study of cryotherapy versus cryotherapy and podophyllotoxin cream as treatment for external anogenital warts. Sex Transm Infect 2009; 85: 514– 519.
- 72 , , et al. 5% Imiquimod cream for external anogenital warts in HIV‐infected patients under HAART therapy. Int J STD AIDS 2004; 15: 17– 20.
- 73 , , et al. Recurrent respiratory papillomatosis: A state‐of‐the‐art review. Respir Med 2017; 126: 116– 121.
- 74 , , et al. Effects of nicotine on the immune response. II. Chronic nicotine treatment induces T cell anergy. J Immunol 1996; 156: 2384– 2390.
- 75 , , . Young women's experiences of managing self‐treatment for anogenital warts. Sex Reprod Healthc 2012; 3: 117– 121.
- 76 , , . Skin infections in pregnancy. Clin Dermatol 2016; 34: 368– 377.
- 77 , , . Imiquimod use in the genital area and development of lichen sclerosus and lichen planus. Int J STD AIDS 2010; 21: 219– 221.
- 78 , , . Recurrent respiratory papillomatosis. Otolaryngol Clin North Am 2012; 45: 671– 694, viii‐ix.
- 79 , . Topical imiquimod: a review of its use in the management of anogenital warts, actinic keratoses, basal cell carcinoma and other skin lesions. Drugs 2007; 67:2187– 2210.
- 80 , , et al. Diabetes and genital warts: an unhappy coalition. Int J STD AIDS 2010; 21: 457– 459.
- 81 , , et al. Four year efficacy of prophylactic human papillomavirus quadrivalent vaccine against low grade cervical, vulvar, and vaginal intraepithelial neoplasia and anogenital warts: randomised controlled trial. BMJ 2010; 341: c3493.
- 82 , , et al. Quadrivalent vaccine against human papillomavirus to prevent anogenital diseases. N Engl J Med 2007; 356: 1928– 1943.
- 83 , , et al. Quadrivalent human papillomavirus vaccination and trends in genital warts in Australia: analysis of national sentinel surveillance data. Lancet Infect Dis2011; 11: 39– 44.
- 84 , , et al. Rapid decline in presentations of genital warts after the implementation of a national quadrivalent human papillomavirus vaccination programme for young women. Sex Transm Infect 2009; 85: 499– 502.
- 85 , . HPV vaccination–reaping the rewards of the appliance of science. BMJ2013; 346: f2184.
- 86 , , et al. Efficacy of quadrivalent HPV vaccine against HPV Infection and disease in males. N Engl J Med 2011; 364: 401– 411.
- 87 , , et al. Recurring infection with ecologically distinct HPV types can explain high prevalence and diversity. Proc Natl Acad Sci U S A 2017; 114: 13573– 13578.
- 88 , , et al. Association of condom use, sexual behaviors, and sexually transmitted infections with the duration of genital human papillomavirus infection among adolescent women. Arch Pediatr Adolesc Med 2006; 160: 151– 156.
Δεν υπάρχουν σχόλια:
Δημοσίευση σχολίου